Surface Plasmon Resonance (SPR) plays a vital role across drug discovery and development, from early candidate screening to detailed product characterisation, including antigen and Fc‑receptor interactions. For biosimilar development, ensuring that SPR methods used for comparative binding analyses are rigorously qualified is essential for regulatory acceptance. This research poster outlines the qualification of multiple SPR assays designed to measure Fcγ receptor, FcRn, and antigen binding to a monoclonal antibody.

|
March 24, 2026
|
4 min read
Throughout the drug discovery and development process, Surface Plasmon Resonance (SPR) has a range of applications from early candidate selection through to product characterisation such as antigen and Fc-receptor binding. When utilising SPR for the analysis of comparative binding during the development of biosimilars, it is important to ensure that any method used for filing to regulatory agencies is carefully qualified for the intended purpose. Herein, we describe the qualification of multiple SPR binding methods for measuring Fcy receptor, FcRn and antigen binding to a monoclonal antibody.
Assays were developed and qualified using a Biacore 8K. Human Fcy receptors and FcRn were expressed and purified in house to enable a design where the antibody test sample is captured in an optimal orientation via the light chain in the Fab region, and the Fc receptors are flowed over the captured antibody in the mobile phase. This has the advantage of reduced avidity effects and negates issues associated with product formulation.
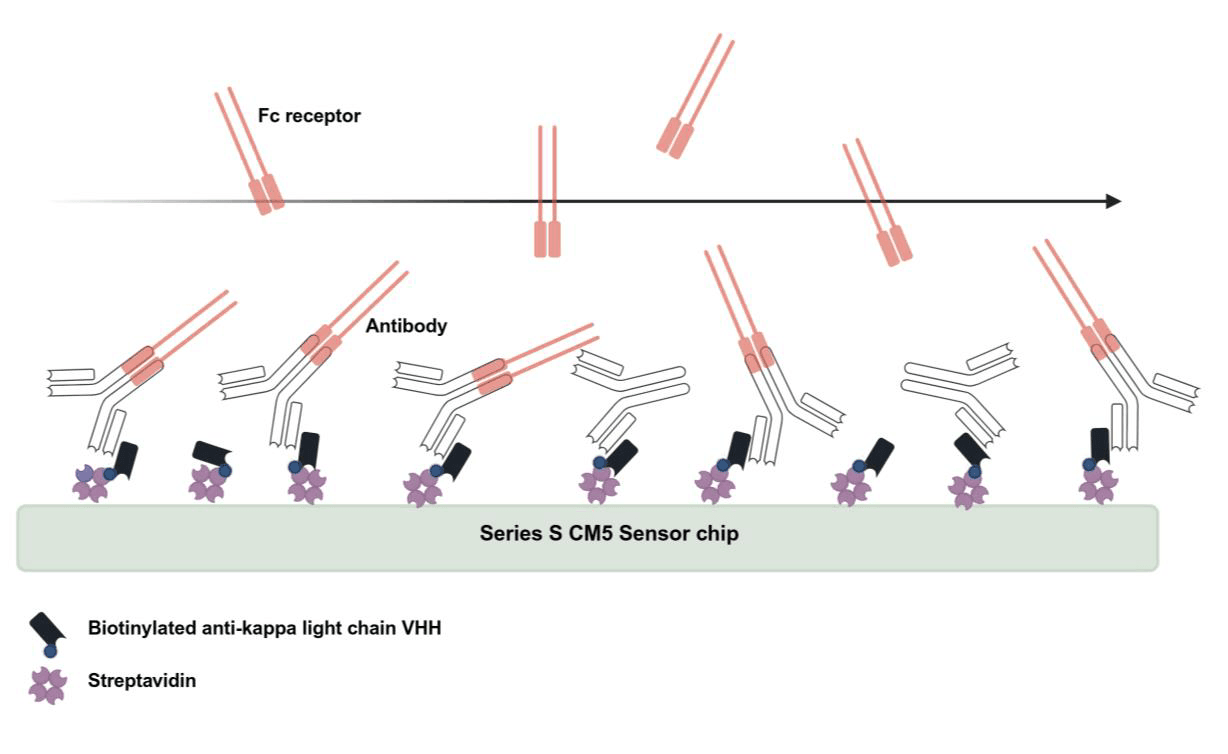
Maintain control of critical reagents by choosing reagents with consistent quality (e.g. purity and activity) and supply. If possible, include multiple batches in qualification assessments.
Assay qualification is performed to demonstrate that an analytical procedure is suitable for the intended purpose, for example product characterisation and comparability assessments.
The following qualification assessments were selected for these methods based on ICH
guideline Q2(R2) on validation of analytical procedures:
• Accuracy
• Precision (including repeatability and intermediate precision)
• Specificity
• Robustness
Eight assay methodologies were successfully developed and qualified using a designated anti-Her2 (IgG1) reference standard (RS), including seven Fcy receptor binding assays, FcRn and target (Her2) binding assays.
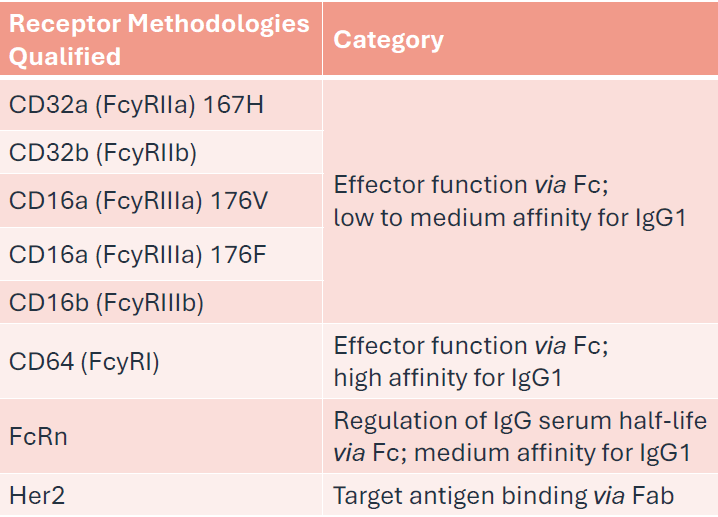
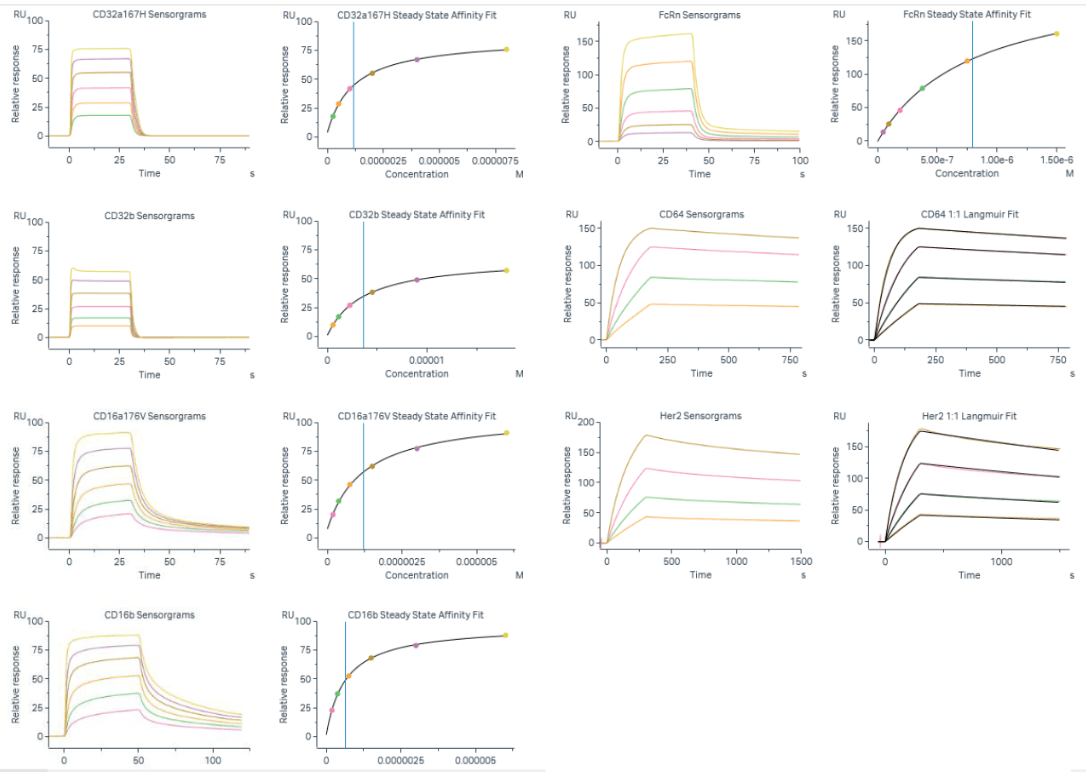
Ensuring that data fits are as optimal as possible during the assay development stages is critical and will influence accuracy and precision results.
For each interaction, 27 assessments of accuracy were performed across 3 assay runs assessing 100% samples prepared from the reference material. All assays demonstrated high accuracy over multiple assessments.
Taking CD64 (FcyRI) as a representative example, accuracy for the absolute KD, ka and kd was calculated relative to the respective mean value of all 27 accuracy assessments. In contrast, accuracy results for the relative values were calculated proportional to the reference standard (RS) material tested within the same assay run.
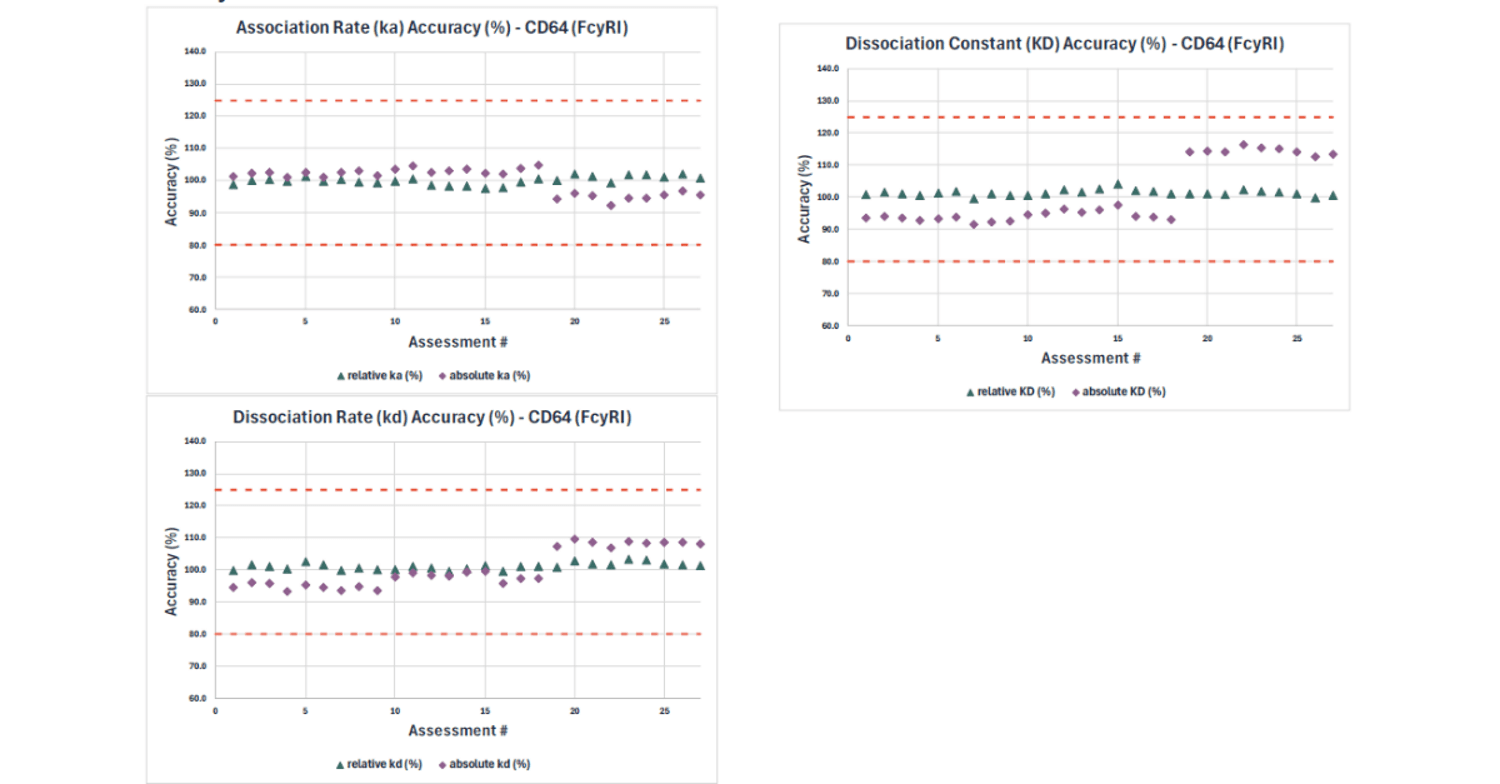
Results for absolute and relative kinetic and affinity values passed the expected qualification pass criterion of 80 – 125% accuracy (represented by dotted lines in graphs).
Absolute values (e.g. KD, ka, kd) were more sensitive to day-to-day variation compared to relative values (expressed as a % of the RS tested within the same run). This is an important consideration for long term assay use, and control of critical reagents and system suitability criteria should be carefully assessed.
Both intermediate precision (n=9) and repeatability (n=3) were assessed and passed the expected qualification criterion of %CV ≤ 20%. The higher variation in intermediate precision observed for absolute affinity values was in line with effects on absolute values that can be caused by day-to-day variation as previously observed for accuracy. assessments.
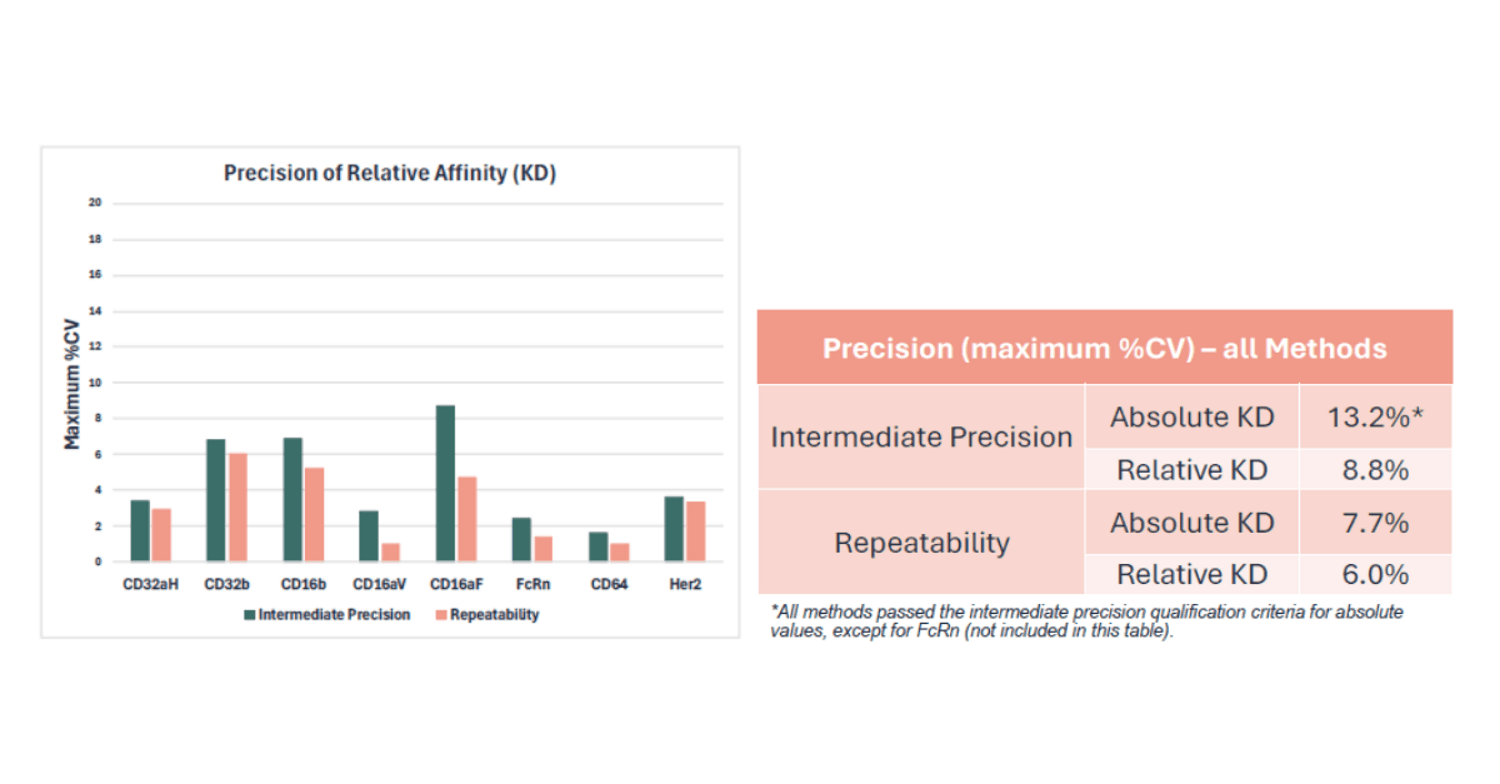
Dilutional linearity and range assessments generally do not apply for kinetic and affinity assays. The reportable value (KD) is a constant which does not vary with dose and analyte concentrations assessed are typically included in a global fitting analysis. Additionally, in this particular assay design, the test sample is captured as the ligand.
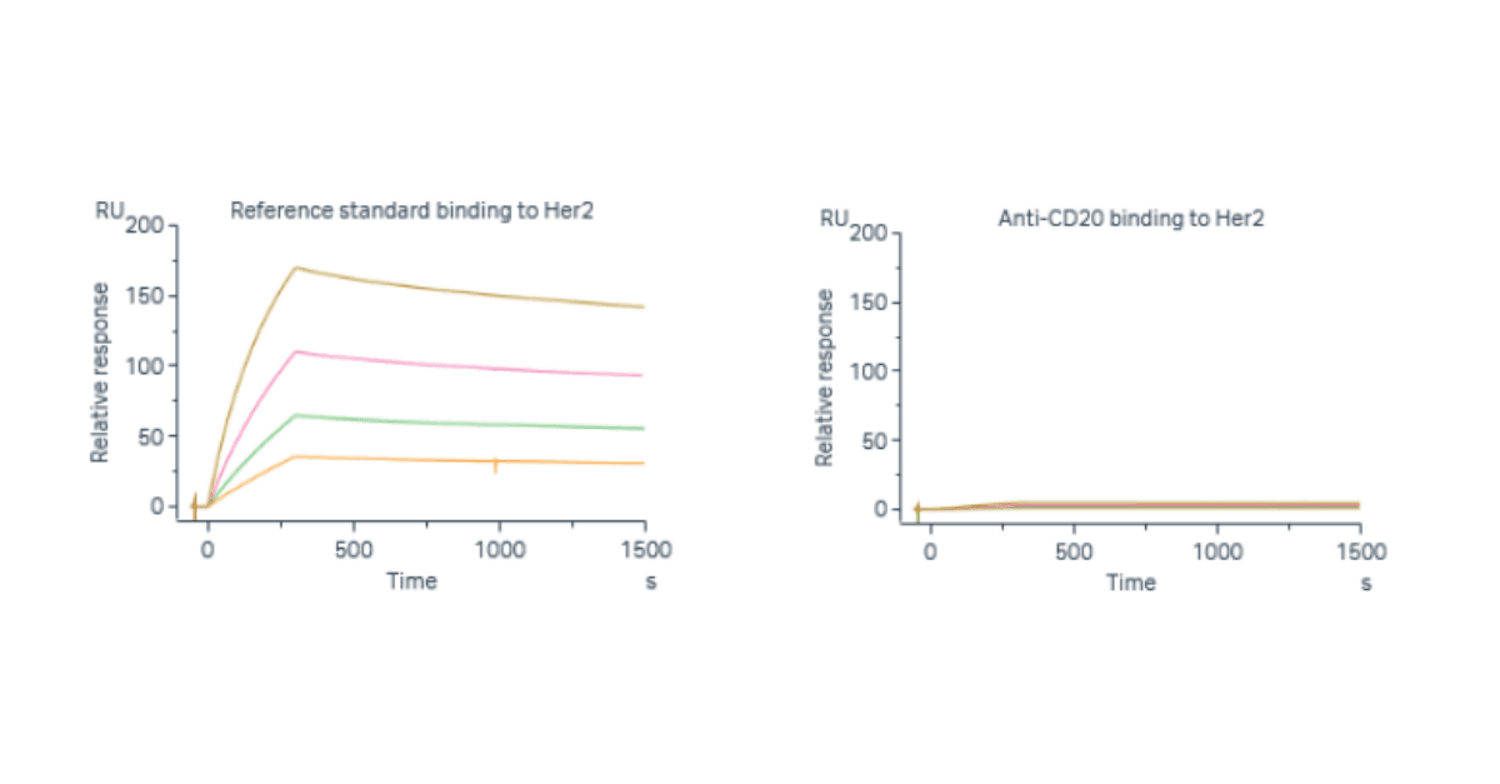
Specificity was assessed for each interaction to demonstrate that the response is specific to the sample of interest and not to structurally related samples. In the example shown here, specificity of binding to Her2 was demonstrated using an anti-CD20 IgG1 specificity control. The qualification criterion of ≤ 20% RU response of a specificity control in comparison to reference material was satisfied for all interactions.
As an indication of reliability during normal usage, a range of robustness assessments were performed. Assay accuracy and precision remained unaffected as values passed all qualification targets.
• Variation of antibody capture concentration between 80-125%
• Analyte reagent lot/batch number
• Extended chip and assay plate storage
Choose a specificity control that is as structurally similar to the sample of interest as possible. For Fcy receptors, an IgG1 control containing point mutations L234S/L235T/G236R in the Fc region that abrogate all Fcy receptor binding (Wilkinson, I et al. (2021) PLoS ONE 16(12)), is routinely used at RoukenBio.
Herein we present the successful qualification of a number of high precision and accuracy SPR-based assays to support biosimilar characterisation. We have demonstrated how ICH guideline Q2(R2) can successfully be applied to kinetics and affinity assays and discuss the resulting considerations for experimental and qualification design. This understanding is important for the increasing emergence of kinetics and affinity assays for successful product characterisation and biosimilar similarity assessments.
🗓️ Stay informed with our monthly scientific newsletter, published on LinkedIn on the last Wednesday of each month.
These editions bring you the latest in drug development breakthroughs, industry trends, and expert insights from the brilliant minds at RoukenBio.
Subscribe today on LinkedInDownload our SPR technical presentation to learn more about all our SPR capabilities and chat to our experts to see how we could collaborate and enhance your drug discovery journey.
Access the technical presentation
