RoukenBio is a specialist immunology CRO providing advanced human in vitro allergy assay development and testing services to accelerate biologics, cell therapy and small molecules. We design mechanism-driven, translational assays that generate decision-grade data for preclinical and early clinical development.
Our advanced human in vitro allergy assay platforms model core type 2 inflammatory pathways, with a strong focus on IgE/FcεRI signalling, mast cell and basophil activation, eosinophil function, Th2 cytokine biology (IL-4, IL-5, IL-13), B-cell class switching, and immune cell cross-talk.
We design and validate allergy-focused cellular assays using primary human immune cells, physiologically relevant co-culture systems, and engineered reporter platforms to deliver high-content, translational data. Our systems incorporate key effector and regulatory cell types driving allergic inflammation, including mast cells, basophils, eosinophils, T helper 2 (Th2) cells, regulatory T cells (Tregs), and B cells.
These platforms support allergy drug discovery and preclinical development by enabling robust assessment of target engagement, potency, mechanism of action (MoA), IgE-mediated activation, cytokine release, degranulation, antibody-dependent cellular responses, and safety-relevant immune function, helping to de-risk next-generation biologics, cell therapies, and small molecule therapeutics.

Allergic disease is driven by inappropriate immune responses to otherwise harmless antigens, often mediated by IgE binding to FcεRI on mast cells and basophils and the release of inflammatory mediators following allergen exposure. As an allergy CRO, we translate this biology into robust, human-relevant allergy assay formats that support allergy drug discovery and candidate progression - especially for therapies targeting IgE pathways, type 2 cytokines (IL-4/IL-13/IL-5), epithelial alarmins (e.g., TSLP, IL-33 and IL-25) and downstream effector cell responses.
Our advanced immunological & bioanalytical techniques help identify and analyse small molecule compounds and antibodies, which are essential in regulating biological processes. By leveraging cutting-edge technology and scientific expertise, we aim to uncover new therapeutic targets and pave the way for innovative treatments.
Explore the diverse range of cell types and assays that RoukenBio offers focusing on allergy research.
Mast cells
Eosinophils
Th2 cells
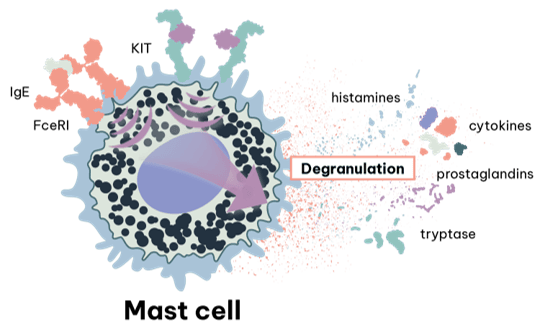
Mast cells are central to many allergy assays because IgE crosslinking at FcεRI triggers rapid degranulation and mediator release (e.g., histamine, proteases, lipid mediators and cytokines). For allergy drug discovery, mast cell models enable direct evaluation of therapies designed to inhibit FcεRI signalling, mediator release and activation pathways.
A central event in acute allergic reactions is the triggering of mast cells, granulated-tissue-resident innate cells found in peripheral tissues such as skin, airways and gastrointestinal tract.
Antigen-specific activation of these mast cells occurs when surface bound IgE is crosslinked by binding of an allergen, driving activation of the mast cell via the high affinity IgE receptor (FεRI). Triggering in this way results in mast cell release of a wide range of proinflammatory and vasoactive mediators derived from preformed granules, arachidonic acid metabolites and as cytokines.
Key examples include histamine, proteases (e.g., tryptase, chymase) and enzymes (e.g., betahexoaminidase); prostaglandins and leukotrienes (e.g., PGD2, LTC4 respectively); and cytokines (e.g., IL-1β, IL-6). This ability to release a wide range of mediators, makes them key players in modulating the immune response and therefore mast cells and their products are attractive therapeutic targets.
A significant barrier to studying human mast cell activity arises from their tissue resident nature, making acquiring sufficient cell numbers for assay use challenging. RoukenBio offers in vitro mast cell differentiation as a valuable tool for use in functional assays for the screening of candidate therapeutics, as well as custom cell lines to monitor triggering of FεRI.

Mast cells are central to many allergy assays because IgE crosslinking at FcεRI triggers rapid degranulation and mediator release (e.g., histamine, proteases, lipid mediators and cytokines). For allergy drug discovery, mast cell models enable direct evaluation of therapies designed to inhibit FcεRI signalling, mediator release and activation pathways.
A central event in acute allergic reactions is the triggering of mast cells, granulated-tissue-resident innate cells found in peripheral tissues such as skin, airways and gastrointestinal tract.
Antigen-specific activation of these mast cells occurs when surface bound IgE is crosslinked by binding of an allergen, driving activation of the mast cell via the high affinity IgE receptor (FεRI). Triggering in this way results in mast cell release of a wide range of proinflammatory and vasoactive mediators derived from preformed granules, arachidonic acid metabolites and as cytokines.
Key examples include histamine, proteases (e.g., tryptase, chymase) and enzymes (e.g., betahexoaminidase); prostaglandins and leukotrienes (e.g., PGD2, LTC4 respectively); and cytokines (e.g., IL-1β, IL-6). This ability to release a wide range of mediators, makes them key players in modulating the immune response and therefore mast cells and their products are attractive therapeutic targets.
A significant barrier to studying human mast cell activity arises from their tissue resident nature, making acquiring sufficient cell numbers for assay use challenging. RoukenBio offers in vitro mast cell differentiation as a valuable tool for use in functional assays for the screening of candidate therapeutics, as well as custom cell lines to monitor triggering of FεRI.
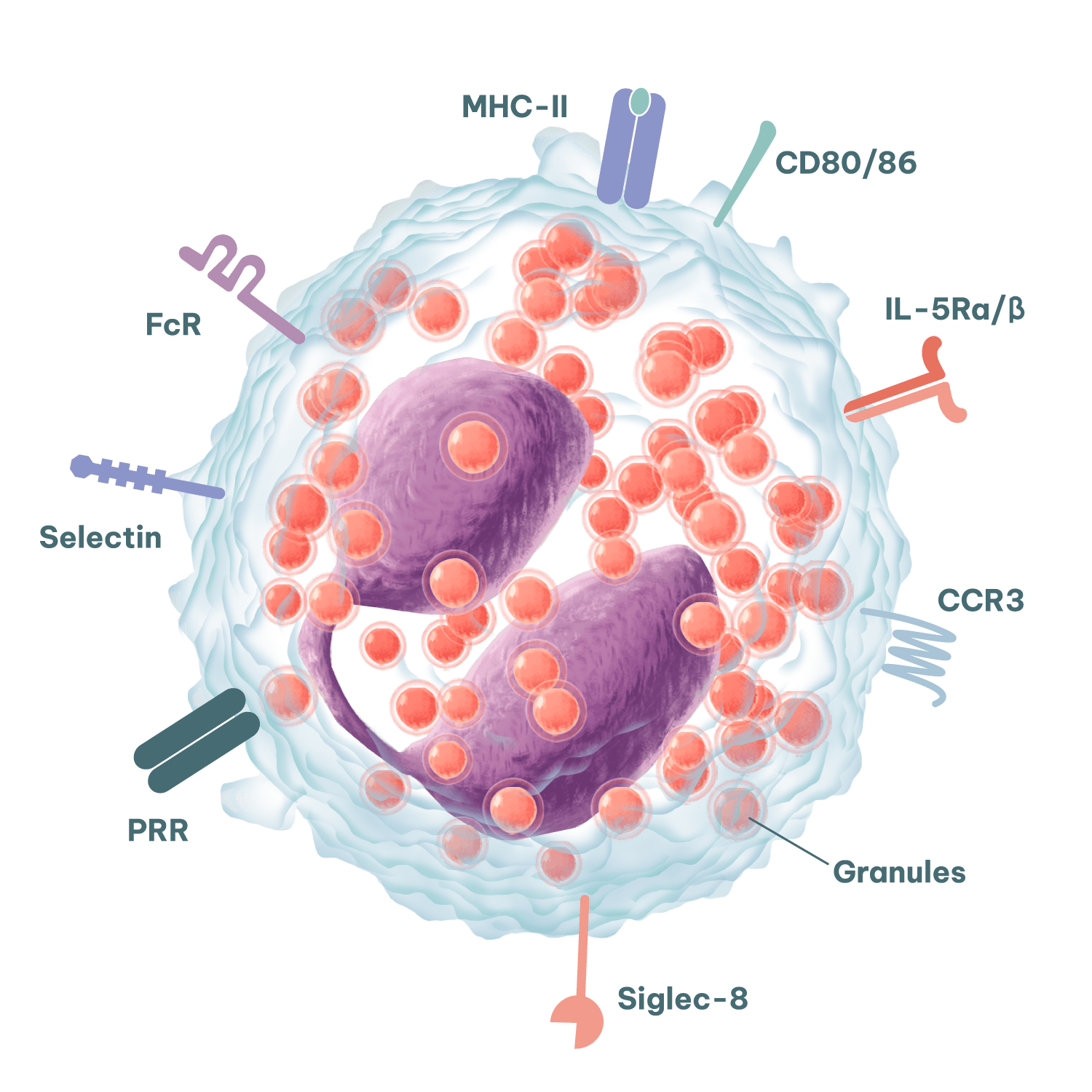
Eosinophils are key drivers of tissue inflammation in allergic disease, making them highly relevant for allergy assay design and profiling of immune-modulating therapeutics.
Upon activation, eosinophils release a host of cytotoxic proteins including major basic protein (MBP), eosinophil cationic protein (ECP), eosinophil-derived neurotoxin (EDN) and eosinophil peroxidase (EPO). In addition, eosinophils can release a host of immunomodulatory cytokines and chemokines to shape adaptive immune responses.
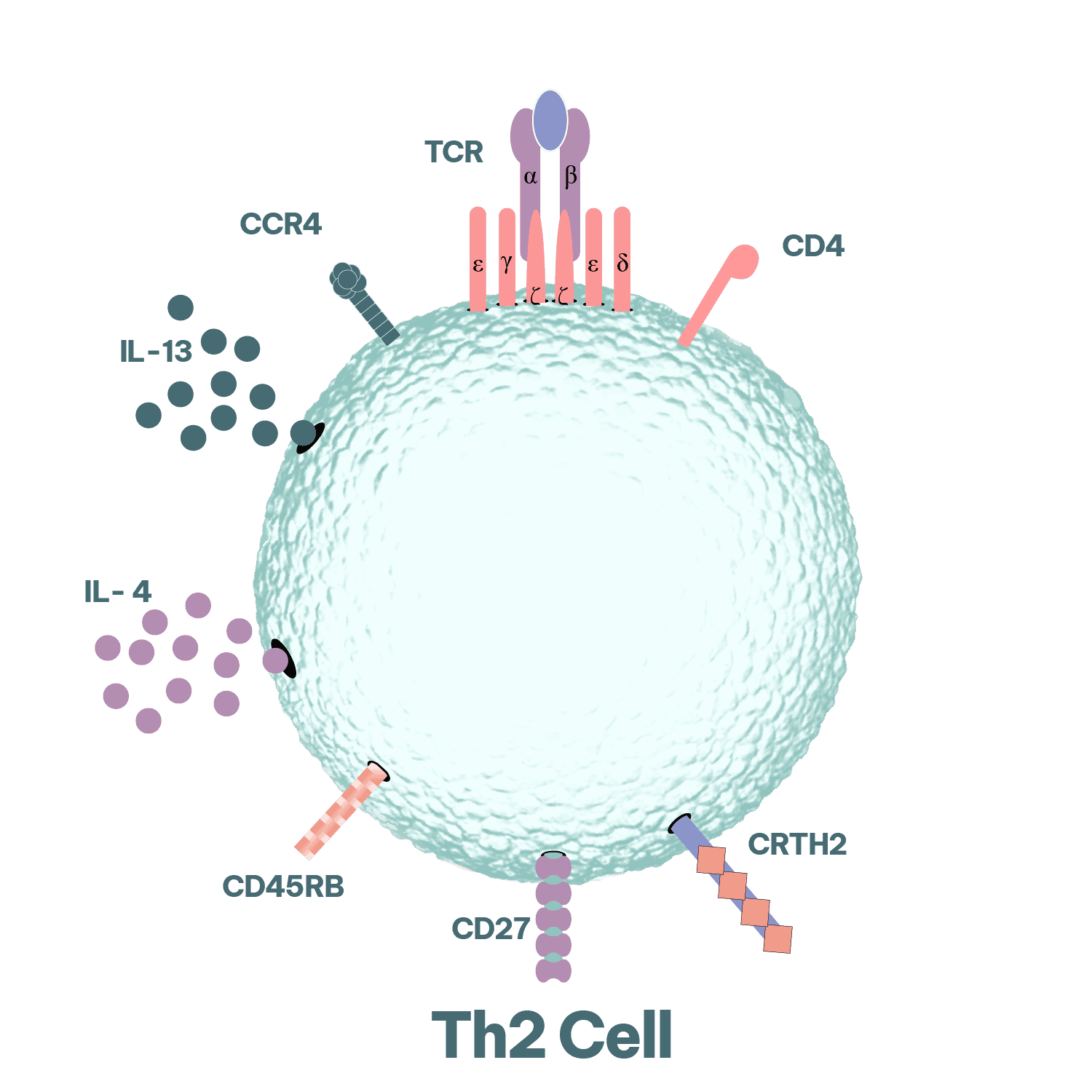
Th2 biology is central to type 2 inflammation and the induction of IgE class switching in B cells. Accordingly, Th2-relevant functional readouts are widely applied in allergy drug discovery to quantify cytokine modulation and to characterise downstream immunological effects on effector cell activation, antibody production, and inflammatory signalling pathways.
CD4 T helper (Th cells) choreograph adaptive immune responses. Th2 cells are associated with helminth infections, however their activity also underlies the inappropriate immune responses seen in allergy. Their activation and secretion of typical Th2 cytokines (IL-4, IL-5, IL-9 and IL-13) drive B cell proliferation and immunoglobulin class-switching to IgE, eosinophilia and mastocytosis, goblet cell hyperplasia, alternative macrophage activation (M2 polarisation) and smooth muscle contraction in type 2 immune responses. Discover more on our T cell assays.
Our allergy therapeutic development services include custom assay development and execution using primary human immune cells, differentiated cell systems and engineered reporter platforms, configured to your modality and mechanism.
Primary mast cell differentiation
Primary eosinophil isolation
Functional allergy assays
Reporter cell lines
h-Differentiation (CD34+): Human CD34+ cells isolated from healthy donor blood and cultured for up to 8 weeks in the presence of recombinant hIL-3, hSCF and hIL-6. Immunophenotyping of the enriched population is performed by flow cytometry for classical surface differentiation biomarkers (e.g., c-Kit/CD117, FεRI) or activation biomarkers (e.g., CD63) upon in vitro activation.
h-Differentiation (CD34+): Human CD34+ cells isolated from healthy donor blood and cultured for up to 8 weeks in the presence of recombinant hIL-3, hSCF and hIL-6. Immunophenotyping of the enriched population is performed by flow cytometry for classical surface differentiation biomarkers (e.g., c-Kit/CD117, FεRI) or activation biomarkers (e.g., CD63) upon in vitro activation.
Freshly isolated from whole blood: Our on-site healthy blood donors allow ready access to human eosinophils isolated directly from freshly drawn blood. The highly enriched population can be immunophenotyped by flow cytometry for a variety of classical biomarkers (e.g. CCR3, IL-5Ra, CD49d, CD15, Siglec-8) and used in custom bioassays.
Mast cell degranulation assay: Mature mast cells can be sensitized with IgE and mast cell activation/degranulation achieved in an antigen driven manner or by employing anti-IgE-antibody. Assay readouts can include measurement of soluble mediators (e.g., tryptase, histamine) by MSD or ELISA or surface biomarkers (e.g., CD63) by flow cytometry.
This list is not exhaustive; if you don’t see the exact allergy assay you need, our specialist team can develop the best assay to answer your questions. Give us a problem, and we’ll find your solution.
Allergy programmes succeed when assays reflect human disease mechanisms and deliver reproducible, interpretable data. RoukenBio provides allergy therapeutic development services that combine translational immunology expertise with advanced primary cell work and engineered cellular tools, so you can progress faster through allergy drug discovery and candidate selection.
From the primary mast cell differentiation to functional assays, our in-house experts and novel solutions are here to guide you through the drug development pipeline.
We leverage our in-depth knowledge of immune cell function to provide reliable and reproducible results.
Tailored solutions to meet specific research needs in allergy and IgE-targeted therapy development.
Access to advanced flow cytometry, functional bioassays, and reporter-based systems.
Let us help accelerate your allergy research and therapeutic development. Contact us today to discuss how our bioassay expertise can support your next breakthrough in allergy treatment.
Access our Capabilities brochure

Visual resources: Selected scientific illustrations on this website were created with BioRender.com and incorporate licensed BioRender content.
An allergy assay is a laboratory test that measures immune responses involved in allergic inflammation, such as IgE/FcεRI signalling, mast cell and basophil activation, Th2 cytokine release, and B-cell class switching. These assays are used in preclinical and early clinical drug discovery to evaluate mechanism of action, potency, and safety of biologics, cell therapies, and small molecules targeting allergic diseases.