RoukenBio delivers human-relevant immunogenicity assay services to support immunogenicity risk assessment for protein-based biotherapeutics. We use in vitro immune models to understand whether a candidate can activate antigen-presenting cells, drive T cell responses, or trigger unsafe cytokine release, helping you de-risk programmes from discovery through IND-enabling stages.
Our portfolio includes dendritic cell maturation assays, t cell functional assay, cytokine release assay (CRA) studies, on-target/off-tumour risk assessment (IndEx-2), and anti-drug antibody (ADA) ELISA workflows.

Immunogenicity, a key safety consideration, is influenced by factors such as treatment methods (e.g., route of administration, therapy duration and frequency), patient characteristics (e.g., immune status, genetic background, disease state) and drug properties (e.g., immunogenic peptide sequences, glycosylation patterns and impurities).
Immunogenic potential can be evaluated using in silico models, MHC-associated peptide proteomics (MAPPs) assay, and various in vitro immune cell assays. These methods identify T cell epitopes and are critical for optimising lead molecules.
Effective immunogenicity risk assessment integrates appropriate assay selection with a representative donor strategy, relevant benchmarks, and predefined decision criteria. RoukenBio designs assay strategies to de‑risk clinical immunogenicity, using complementary in vitro systems to interrogate the drivers of anti‑drug antibody responses in the context of the intended patient population.
DC Maturation Assays
T Cell Functional Assays
Cytokine Release Assay (CRA)
On-target, off-tumour assessment with IndEx-2
ADA ELISA
Dendritic cells (DCs) are key antigen-presenting cells. Assessing their maturation by monitoring cell surface marker expression (e.g., CD83, CD80, CD86, CD40), cytokine secretion (e.g., IL-1β, IL-6, IL-12, TNFα) or signalling pathway activation (e.g., Akt, Syk) via flow cytometry or ELISA provides insights into the immunogenic potential of biotherapeutics. These assays use monocyte-derived dendritic cells (moDCs), differentiated from PBMCs with GM-CSF and IL-4, to evaluate biotherapeutic-induced activation.
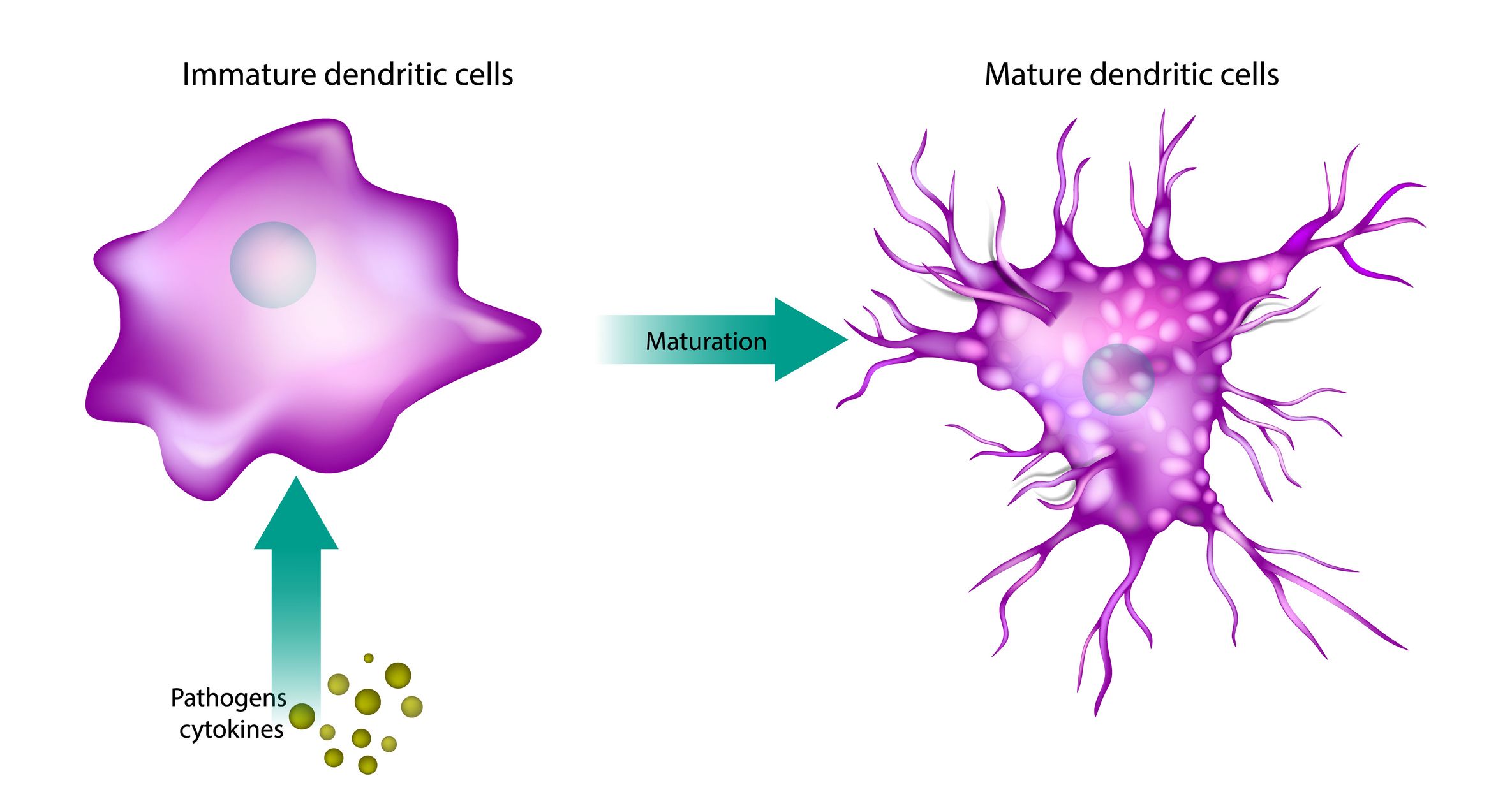
Dendritic cells (DCs) are key antigen-presenting cells. Assessing their maturation by monitoring cell surface marker expression (e.g., CD83, CD80, CD86, CD40), cytokine secretion (e.g., IL-1β, IL-6, IL-12, TNFα) or signalling pathway activation (e.g., Akt, Syk) via flow cytometry or ELISA provides insights into the immunogenic potential of biotherapeutics. These assays use monocyte-derived dendritic cells (moDCs), differentiated from PBMCs with GM-CSF and IL-4, to evaluate biotherapeutic-induced activation.

Our T cell activation assay formats measure CD4+ T cell responses to therapeutic proteins, predicting immunogenic risk. Assay setups include PBMC-based or DC-T cell co-cultures, with readouts such as activation markers (CD154, CD134, CD137), cytokine secretion (IL-2, IFNγ, TNFα), and T cell proliferation (proliferation dyes, Ki-67 expression), key functional endpoints in immunogenicity risk assessment.
Controls like KLH and benchmark drugs (e.g., bococizumab, ATR-107 and bevacizumab) ensure assay validity and comparative analysis.
An in vitro cytokine release assay (CRA) predict cytokine release from immune cells exposed to biotherapeutics, aiding preclinical toxicology assessments. The basic principle of CRAs is incubation of immune cells with the biotherapeutic over a defined period followed by assessment of cytokine release usually with a multiplex approach (e.g., Luminex or MSD). Formats vary by immune cell source (whole blood or PBMCs) and biotherapeutic application (soluble or immobilised).
We often use a tiered CRA approach, screening across formats in a smaller donor set, then confirming in the most relevant format across a wider donor panel.
Important assay considerations include:
The IndEx-2 system evaluates antigen density and activation thresholds to address on-target, off-tumour side effects in antibody-targeted therapies. This system controls the expression of one or two proteins over a dynamic range, linking antigen density to activation thresholds for cytolysis and cytokine release. This innovative approach supports screening, characterisation, and lead selection for mono- and dual-targeted therapies. At RoukenBio, we embrace change and drive innovation, exemplified by our trademarked IndEx-2 system.
Anti-Drug Antibody (ADA) ELISA is essential for detecting immune responses to biotherapeutics. This assay identifies and quantifies antibodies against therapeutic proteins, providing insights into immunogenicity thereby aiding in risk management and ensuring therapeutic efficacy and safety. Formats include screening assays to identify the presence of ADA, confirmatory assays to validate ADA specificity and neutralising assays to assess the inhibition of therapeutic protein activity by ADA.
See how our dual inducible cell line system, combined with primary human cell assays, helps quantify activation thresholds and evaluate on-target/off-tumour risk, so you can make clearer go/no-go decisions.
WATCH THE WEBINAR

An immunogenicity assay measures the ability of a biologic to induce an immune response, typically by measuring CD4 T cell activation or proliferation, which act as a surrogate for anti-drug antibody generation. The magnitude and nature of this response is used to assess potential impacts on safety and efficacy.